A majority of Americans support greater access to experimental drugs before clinical trials have been completed, according to a recent Pew Research Center report. But there are notable differences in opinion on this issue by income, education and race.
Generally, adults from higher-income households and college degree earners are more likely than others to favor greater availability of investigative medical treatments. And, when it comes to different racial groups, African-Americans are significantly less supportive of the idea than whites and Hispanics.
The U.S. Food and Drug Administration recently announced a more streamlined process for doctors to obtain experimental drugs for seriously ill patients. The proposed changes are expected to speed up the application process and get investigative treatments to patients more quickly and efficiently.
The new FDA guidelines come after five states – Arizona, Colorado, Louisiana, Michigan and Missouri – passed legislation in 2014 that would give terminally ill patients greater access to medical treatments that have not been fully evaluated by the FDA. At least 25 other states have filed similar “right to try” laws in the past year, and just last week, Virginia lawmakers unanimously passed legislation seeking to expand patients’ access to investigative experimental drugs.
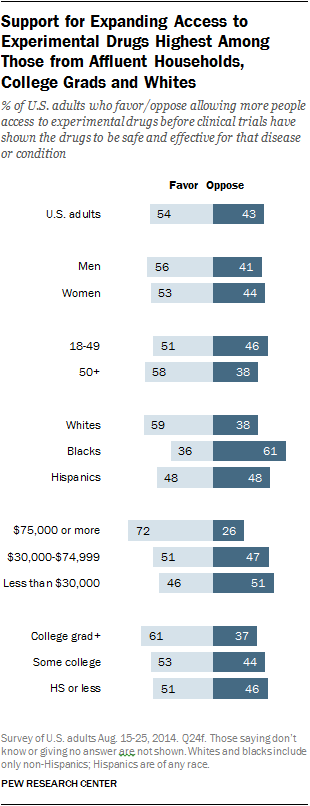
A Pew Research survey from August 2014 asked adults: “All in all, do you favor or oppose allowing more people access to experimental drugs before clinical trials have shown the drugs to be safe and effective for that disease or condition?”
Just over half (54%) of U.S. adults say they were in favor of greater access to experimental drugs before clinical trials have shown drugs to be safe or effective, while 43% oppose wider access. Views, however, vary greatly by household income and race.
Nearly three-quarters (72%) of adults who live in households earning $75,000 or more a year favor allowing more people access to experimental drugs before clinical trials are finished, while 26% are against wider availability of such drugs. In comparison, adults whose annual family income is less than $30,000 are more divided on the issue: 46% are in favor of wider access while 51% are in opposition.
Another big divide in opinion is among racial groups. Support is highest among whites – 59% of whites back greater access to experimental drugs, compared with 48% of Hispanic and only 36% of blacks. Roughly six-in-ten blacks (61%) oppose making experimental drugs more widely available before such drugs have been fully evaluated.
One possible reason for African-Americans’ views on experimental drugs may be tied to the legacy of mistreatment of blacks by the medical establishment over generations, most infamously in the Tuskegee syphilis experiment. Over four decades, beginning in 1932, the U.S. Public Health Service recruited hundreds of black men for medical trials in rural Alabama in which participants were misled about the study’s purpose and were never given proper medical treatment.
Karen Bullock, a professor of medical sociology and social work at North Carolina State University, says the “attitudes of mistrust” displayed by some older black Americans are tied to their “undisputable lived experiences,” in that some may remember when the trials first became public.
Bullock also notes that disparities in health outcomes between blacks and whites may be linked to blacks’ less favorable views of expanding access to experimental drugs, and that another indicator of blacks’ wariness of drug-testing experiments is their relatively low participation rates in clinical trials.
When it comes to other demographic differences, college graduates (61%) are somewhat more inclined to support expanding access to experimental drugs before trials are finished than those with only some college experience (53%) or who have a high school diploma or less (51%). Men and women are about equally likely to favor increased access to experimental drugs before clinical trials are complete, as are those under and over age 50.
